Bonding
In solids, there are three different types of primary or chemical bonding : ionic, covalent, and metallic. The type of bonding depends on the structure of the electrons. There are also secondary forces found in solid materials that, while weaker than the primary bonds, can still influence the physical properties of materials.
Ionic Bonding
The first type of primary bonding is called ionic bonding. It is always found in compounds that contain metallic and nonmetallic elements. This happens because atoms from a metallic element will easily give their valence electrons to nonmetallic atoms, therefore filling the orbital shells and acquiring an electric charge, i.e., becoming ions. An example can be seen below where we see the extra valence elctron if given over to fill the other shell.
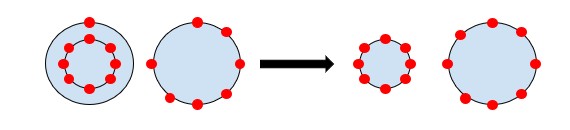
Covalent Bonding
The second type of primary bonding is called covalent bonding. This is found in materials that have small differences in electronegativity or, in other words, sit close to each other on the periodic table. This type of bonding happens when two atoms share electrons. It is very common between nonmetallic elements. An example of this can be seen below:
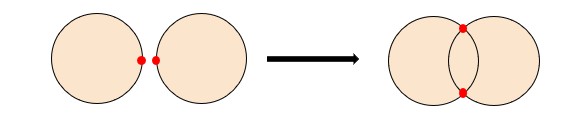
Metallic Bonding
The last type of primary bonding is called metallic bonding, which is found in metals and metal alloys. This occurs when the valence electrons are not bound to a specific atom but instead form a "sea of electrons" that can drift around throughout the metal.
Secondary Bonds (Van der Waals)
Finally, secondary bonds, also known as van der Waals bonds, are bonds that exist in almost all atoms and molecules, though they are often obscured by primary bonding as it is much weaker.
Exercise
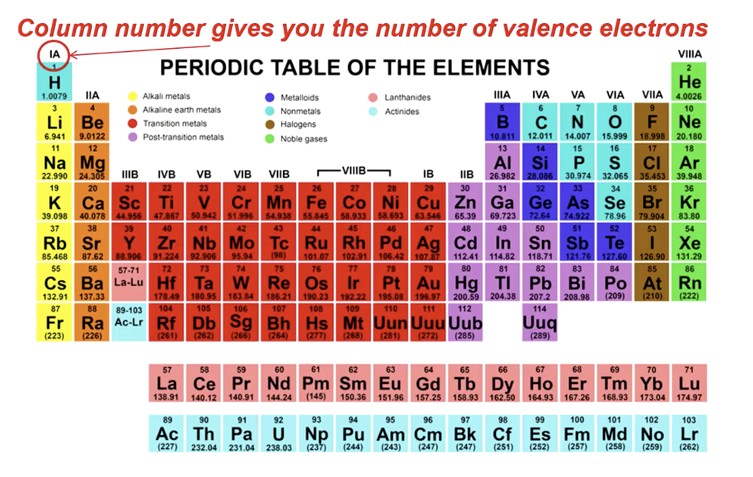
Using the periodic table, identify the dominant bonding type for each material below.
- Brass (copper-zinc alloy)
- Rubber
- Calcium fluoride (CaF\( _2 \))
- Bronze
- Solid xenon
- Tungsten
- Cadmium telluride (CdTe)